
Providing researchers with quality and transparency of FBS
Serum is commonly used as a supplement to basal growth medium in cell culture. The most common type of serum is fetal bovine serum (FBS) because of its high content of embryonic growth promoting factors. In cell culture, serum provides a wide variety of macromolecular proteins, low molecular weight nutrients, carrier proteins for water – insoluble components, and other compounds necessary for in vitro growth of cells, such as hormones and attachment factors. Serum also adds buffering capacity to the medium and binds or neutralizes toxic components. The selection of a serum supplement for cell culture applications is primarily dependent on the chemical definition of the basal medium, the type of cell to be grown, and the culture system being employed. Below is a detailed overview in the collection methods and development of producing high performance FBS.
FBS collection and processing processes
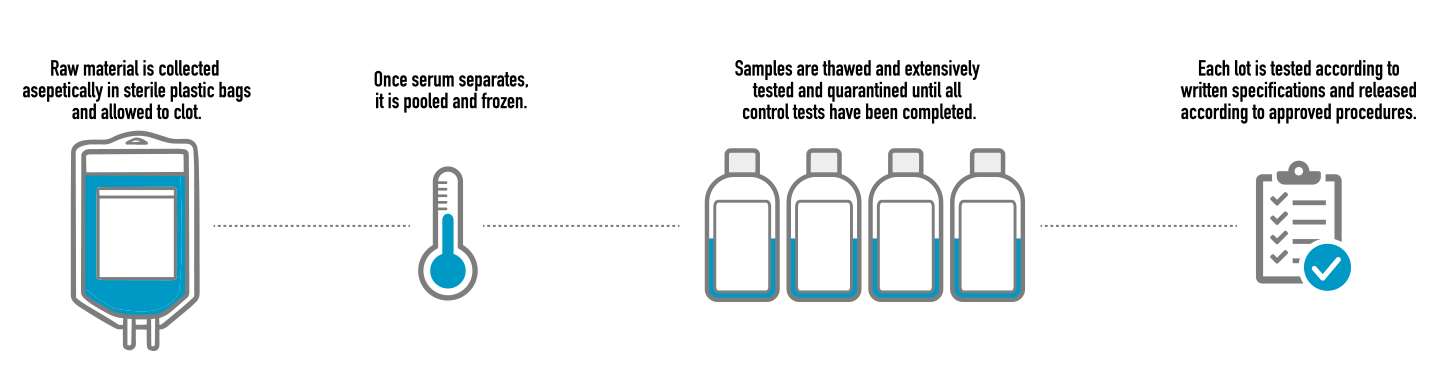
FBS collection process. In the FBS manufacturing process, whole blood is collected aseptically in disposable sterile plastic bags and allowed to clot. Once the serum has been separated from the clot, it is pooled and frozen. Controlling the initial collection is a crucial factor in the quality of the final serum product. Only raw material that meets our specifications is approved for production.
Processing serum samples. Selected batches of serum raw material are thawed, tested for endotoxins and hemoglobin content and only the accepted material is pooled. The pooled raw material is thoroughly blended under refrigerated conditions and membrane filtered for sterility according to a well validated filtration protocol. Biological Industries processes FBS through a sequence of prefilters and membrane filters. The filtration step includes the use of three 0.1 micron sterilizing grade membrane filters in series.
After filtration, the serum is dispensed into bottles by an aseptic filling process which has been validated to insure sterility of the final product. Serum products are produced in a controlled environment (clean rooms) designed to carefully control air pressure and particulate matter. The manufacturing area is a class 100,000 (ISO 8) environment. The sterile bottles and equipment are stored in a class 10,000 (ISO 7) environment, and the filling room is a class 1000 (ISO 6) environment with class 100 (ISO 5) laminar air flow sterile bench. Clean rooms are monitored on a regular basis for particulate and microbial levels to ensure that the air handling system, cleaning protocols, and personnel maintain standards control. After filling, the final product is quickly frozen to -20ºC and held in quarantine until all quality control tests have been completed.
Quality Control
Each lot of FBS is tested to confirm that the serum meets the written specifications. Final product release is done after reviewing all production and quality control records to determine compliance with all established, approved written procedures. Tests include:
Physical and Chemical Tests
- Electrophoretic pattern
- PH
- Osmolality
- Total proteins
- Albumin
- IgG
- Hemoglobin
- Globulins
Biochemical Tests
- Alanine Transaminase (ALT)
- Alkaline Phosphatase
- Aspartate Aminotransferase (ast)
- Bilirubin – total
- Bilirubin – direct
- Blood Urea Nitrogen (BUN)
- Calcium
- Chloride
- Cholesterol
- Creatinine
- Creatinine Kinase (CK)
- Gamma-Glutamyl Transferase (GGT)
- Glucose
- High Density Lipoproteins (HDL)
- Lactate Dehydrogenase
- Low density Lipoproteins (LDL)
- Phosphorous (Inorganic)
- Potassium
- Sodium
- Triglycerides (TG)
- Uric Acid
Microbiological Tests
- Sterility tests: Bacterial and fungal sterility tests according to the current USP.
- Mycoplasma contamination: according to the CFR, title 9, part 113 (culture method).
- Viral contaminants: according to the protocols described in CFR, title 9, part 113 for BVD, IBR and PI3.
- Viral antibodies: screened to determine the titer of neutralizing antibodies to BVD, IBR and PI3.
- Endotoxins: test is performed using kinetic colorimetric method .
Biological performance
Cell growth tests are designed to check the efficacy of the FBS in promoting cell growth. Cells used are fibroblasts (MRC-5 diploid normal cells), epithelial cells (Hep-2) and hybridoma cells. Each test is conducted using the tested serum and a validated control lot. Growth promotion using MRC-5 cells is evaluated through several subculture generations to observe any evidence of cytotoxicity and morphological changes of the cells.
- Hep-2 cells (ATCC, CCL 23): growth curve.
- MRC-5 cells (ATCC, CCL 171): 3 passages test.
- Hybridoma cells: cloning efficiency testing.
Quality assurance - CE mark
Biological Industries FBS production process is carried out under controlled conditions in a controlled environment. The steam-in-place (SIP) sterilization , filtration for sterility and filling are validated as required for key aseptic processes. A dossier (Device Master record) exist for serum with all relevant data concerning serum production. The production process from the raw material to the final product in storage, as well as the quality control tests and results, are documented and filed to ensure traceability and control of the process.
Biological Industries products are manufactured in compliance with the quality management standard ISO 9001:2008 and ISO 13485:2016. In addition, the FBS production process conforms to the In Vitro Diagnostics Directive (IVDD 78/79/EC) of the European Parliament. Therefore, our FBS received the CE mark making it eligible for sale in the European Union for in vitro diagnostics. A Bovine Spongiform Encephalopathy (BSE) certificate of suitability had been issued to Biological Industries by the European Directorate for the Quality of Medicines (EDQM) in accordance with monographs of the European Pharmacopoeia. All documents and certifications are available upon request.
Raw material collected or manufactured by Biological Industries
Biological Industries offers US-origin FBS and USDA-grade FBS. US Origin FBS is FBS produced and manufactured from raw material originating in the United States. USDA-Grade FBS is produced from raw materials originating only from countries certified to be free of both BSE (Bovine Spongiform Encephalopathy) and FMD (Foot and Mouth Disease). USDA-Grade FBS can be freely imported into any country, and both these types are the product choice in all countries for manufacturing purposes. The use of US-origin or USDA-grade allows researchers to send their cells, or the products of their cells, to collaborators in other countries with strict import regulations. All FBS products processed in Biological Industries manufacturing facility in Israel is USDA-Grade FBS.
We have sera qualified for specialty research and specific assays, including stem cell research, immunoassays, antibodies, and more.
Product |
Description details/usage |
|---|---|
GAMMA-IRRADIATED FBS |
Gamma irradiation is a process which involves exposing material to high energy gamma photons released by radioisotopes, such as cobalt 60. This energy is transferred from the photons to the products, and is responsible for organism inactivation by ionization of nucleic acid bonds. The irradiation of serum is intended to provide complete assurance of viral inactivation. An extensive validation study has been performed to validate the irradiation process using FBS spiked with several viruses . We have demonstrated that properties and cell culture performance of FBS are not altered by gamma irradiation exposure up to 3.5 MRad. |
DIALYZED FBS |
Most cells grown in culture require a serum component of the growth medium to maintain their proliferative capacity. While whole serum is permissible for routine purposes, studies involving nutritional parameters or incorporation of labeled material require that the constituent under study be removed from the serum. The most commonly used method for removal of these constituents is dialysis of whole serum. For dialysis by diafiltration, serum is circulated through a hollow-fiber by the concentration method. The filtrate however, is replaced by the addition of physiological saline to the serum. |
HEAT INACTIVATED FBS |
Heat inactivation of serum is performed by raising the temperature of the serum to 56oC and maintaining that temperature for 30 minutes. Heat inactivation is the method of choice to destroy complement, and to ensure that the cells will not be lysed by antibody binding. |
EMBRYONIC STEM CELL TESTED FBS |
Embryonic Stem (ES) Cells are pluripotent cells derived from the inner cell mass of the blastocyst. The stem cells can be maintained in vitro for extended periods without loss of their capacity to differentiate to all cell lineages when reimplanted back into a blastocyst. ES cells may differentiate in vitro to a variety of cell types including neuronal, muscle, endothelial and hemapotoietic progenitors. General culture conditions are well established and usual require ES cells to be grown on an inactive feeder cell layer or with basement membrane components (Matrigel, Fibronectin, Laminin). When growing ES cells, one of the most of the most important parameters is the maintenance of the cells in the undifferentiated state. Pre-screening of the serum is essential before using it for the culture of ES cells. Various lots of sera are screened for the growth of Human ES cells using MEF`s as a feeder layer for four passages. The following parameters are measured for the screening: Human ES cells colony morphology, plating efficiency, and differentiation rate: analysis of human ES cells surface marker expressed on the undifferentiated cells membrane (FACS analysis). The results are used as an indication of the quality of sera for the growth and maintenance of undifferentiated stem cells. |
MESENCHYMAL STEM CELL TESTED FBS |
Human mesenchymal stem cells (hMSC) are multipotent adult stem cells present in a variety of tissue niches in the human body. hMSC have advantages over other stem cell types due to the broad variety of their tissue sources, since they are immuno-privileged, and for their ability to specifically migrate to tumors and wounds in vivo. Mesenchymal stem cell qualified FBS are pre-screened for the following: Ability of stem cell differentiation, morphology, cell growth and expansion, and clonal efficiency. Each lot of FBS is extensively tested for its ability to support in vitro growth and expansion of undifferentiated hMSCs. The results are utilized as an indication of the highest purity of the sera that emphasizes maximum expansion of unaltered MSC's, while at the same time, maintaining their unrestricted multiipotency. |
FUNCTIONALLY TESTED for use with TETRACYCLINE REGULATED SYSTEM FBS |
In these systems, a promoter is switched on or off by tetracycline, enabling control of the expression of a cloned gene. These special lots of FBS have been pre-tested to ensure that they permit the range of tetracycline-regulated induction with well characterized Tet lines. As with all of our FBS products, the raw materials always originates from BSE and FMD-free countries. |
CHARCOAL-STRIPPED FBS |
Charcoal-stripped FBS is used to elucidate the effects of hormones in a variety of in vitro systems. Studies include steroid- receptor binding, steroid regulation of cellular receptors, hormone secretion of various tissues and the function of thyroid hormones. The production procedure includes the use of charcoal and dextran to remove the hormones from the FBS. |
Serum reservation program
Serum, as a biological material, represents an undefined mixture of components in which the fetal bovine serum composition varies from one lot to the other. Some cell types are sensitive to the variations in serum performance. Customers are encouraged to evaluate serum samples with their own culture system and cells while we reserve the quantities of the specific lots until customer testing is completed. To learn more, please feel free to email us at custom-services@bioind.com or send us an online inquiry and a member of our Customer Service team will contact you.





