Description
Details
Product Overview
MSCgo™ Chondrogenic Differentiation Medium is a serum-free (SF) and xeno-free (XF) formulation developed for optimal differentiation of human mesenchymal stem cells (hMSC) to mature chondrocytes. The MSCgo™ Chondrogenic Differentiation Medium is validated to efficiently differeniate hMSC from a variety of sources, including bone marrow (BM-MSC), adipose tissue (AT-MSC), and umbilical cord tissue (UC-MSC).
The MSCgo™ Chondrogenic Differentiation Medium kit includes a basal medium and supplement, containing all growth factors and supplements necessary for a complete chondrogenic differentiation medium. No additional supplements are required.
The MSCgo™ Chondrogenic Differentiation protocol is part of a complete system for multipotency evaluation of hMSCs. This kit enables reliable differentiation of hMSCs to mature chondrocytes without background differentiation or interruption in cellular metabolism.
Features
- Serum-free, xeno-free medium
- All required growth factors and supplements included in kit
- Reliable differentiation to mature chondrocytes
- Each lot is application tested
- Does not contain antibiotics
Components
- MSCgo Chondrogenic Differentiation Basal Medium: 100 mL
- MSCgo Chondrogenic Differentiation Supplement Mix : 10 mL
Chondrogenesis Results
Chondrogenic differentiation of hMSC in 3D spheroid culture in the formation of cartilage with a typical extracellular matrix rich of Aggrecan. Aggrecan is a proteoglycan that can be used as an indicator for cartilage formation and can be detected with Alcian Blue, a dark-blue copper-containing dye that acts as an indication of mature chondrocytes. The staining intensity can be vary using different hMSC (e.g. source, age, and passage number).
Cartilage differentiation results of hMSC from various sources after 21 day assay using MSCgo™ Chondrogenic followed by Alcian Blue staining.
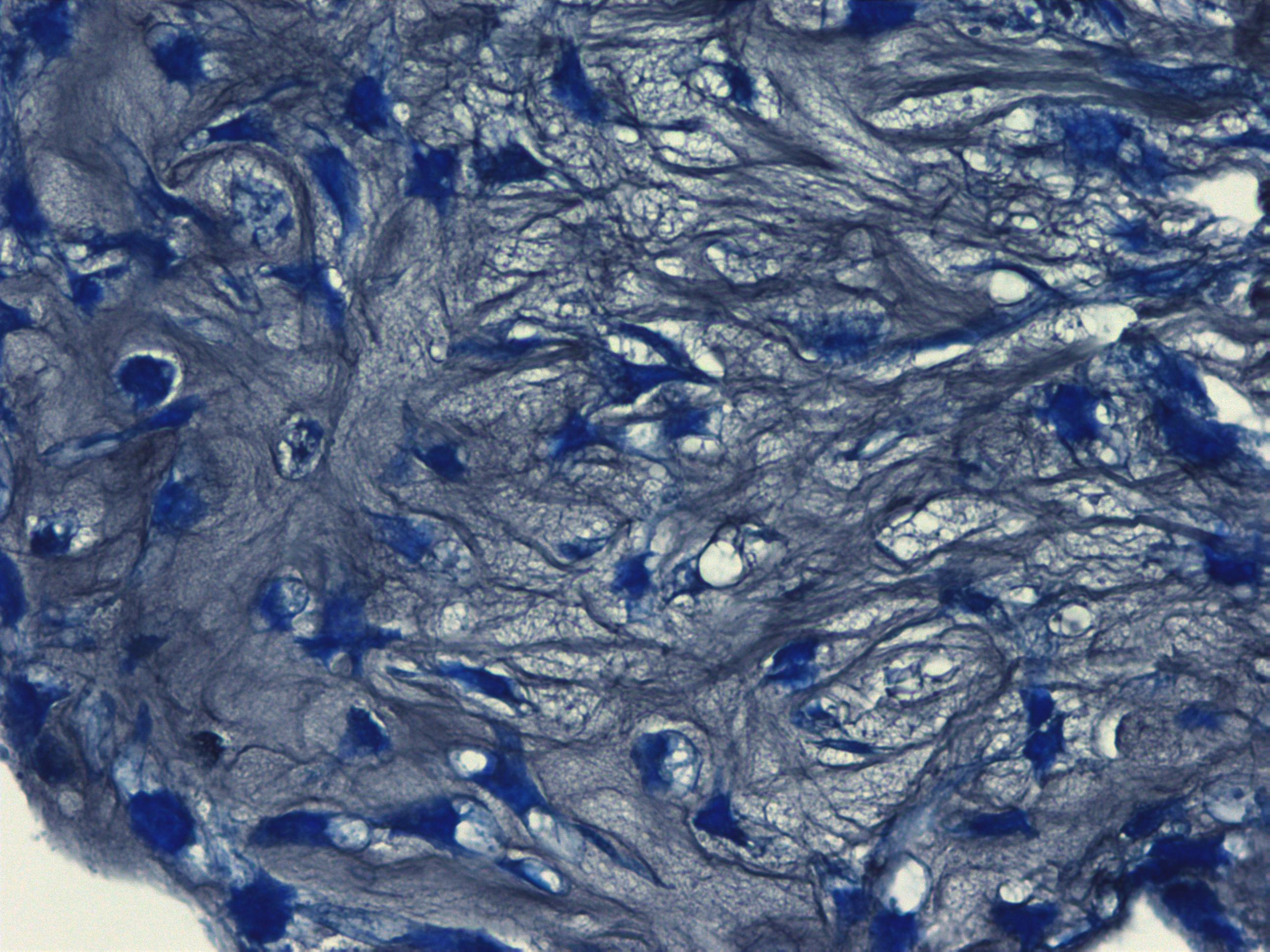
Histological images of mature chondrocytes surrounded by a cartilage matrix after a 21-day differentiation assay using MSCgo™ Chondrogenic Medium followed by Toluidine blue (40x).
Specifications
Specifications
| QTY | 100 mL |
|---|---|
| Form | Liquid |
| Brand | MSCgo™ |
| Storage Conditions | Chondrogenic Differentiation Basal Medium: 2 to 8°C Chondrogenic Differentiation Supplement Mix : -20°C |
| Shipping Conditions | Cold Pack |
| Quality Control | The MSCgo Chondrogenic Differentiation Kit is validated for optimal differentiation of hMSC into chondroocytes. Additional tests are: pH, osmolality, endotoxins and sterility tests. |
| Specifications | Required Materials for Chondrogenic Assay
|
| Instructions for Use | Complete Medium Preparation
Note: The combination of the MSCgo Chondrogenic Supplement Mix into the Basal Medium generates a complete medium that is ready for use. No additional supplements are required. |
| Legal | For human ex vivo tissue and cell culture processing applications. This reagent is not approved for human or animal use, or for application of in vitro diagnostic procedures. |
References
references
- J. Leber et al., Microcarrier choice and bead-to-bead transfer for human mesenchymal stem cells in serum-containing and chemically defined media. Process Biochemistry, volume 59, Part B, August 2017, Pages 255-265
- L. Pu et al., Compared to the amniotic membrane, Wharton’s jelly may be a more suitable source of mesenchymal stem cells for cardiovascular tissue engineering and clinical regeneration. Stem Cell Research & Therapy 2017 8:72
Documentation
Materials Safety Data Sheet
 MSCgo™ Chondrogenic Differentiation Medium (MSDS)
MSCgo™ Chondrogenic Differentiation Medium (MSDS) MSCgo™ Chondrogenic Differentiation Supplement Mix (MSDS)
MSCgo™ Chondrogenic Differentiation Supplement Mix (MSDS)
Manuals and Protocols
Product Literature
Certificate of Analysis
COA's can be downloaded from Sartorius's Certificates Portal.
For certificates issued before November 15, 2021, please enter below the product lot number and click search.